
You are now leaving www.klisyrihcp.com
You are now leaving KlisyriHCP.com and will be directed to an independent, third-party website. The Privacy Policy and the Terms of Use of the destination website will apply.
Quick Links
KLISYRI is a microtubule inhibitor indicated for the topical treatment of actinic keratosis of the face or scalp
KLISYRI may cause eye irritation. Avoid transfer of the drug into the eyes and to the periocular area during and after application. Wash hands immediately after application. If accidental exposure occurs, instruct patient to flush eyes with water and seek medical care as soon as possible.

*
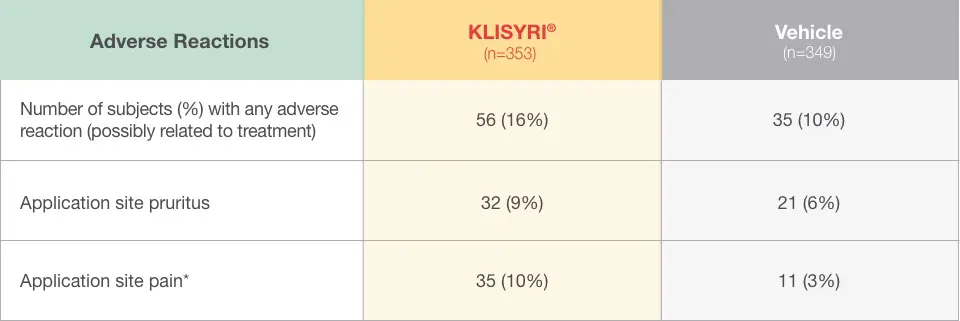
Application site pain includes pain, tenderness, stinging, and burning sensation at the application site.
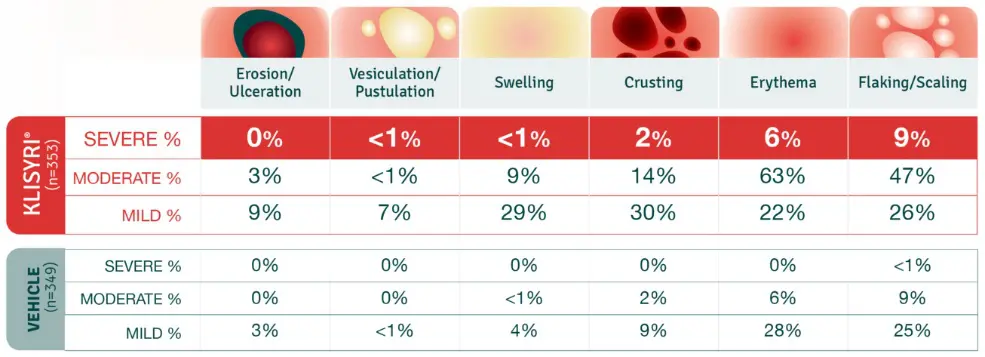
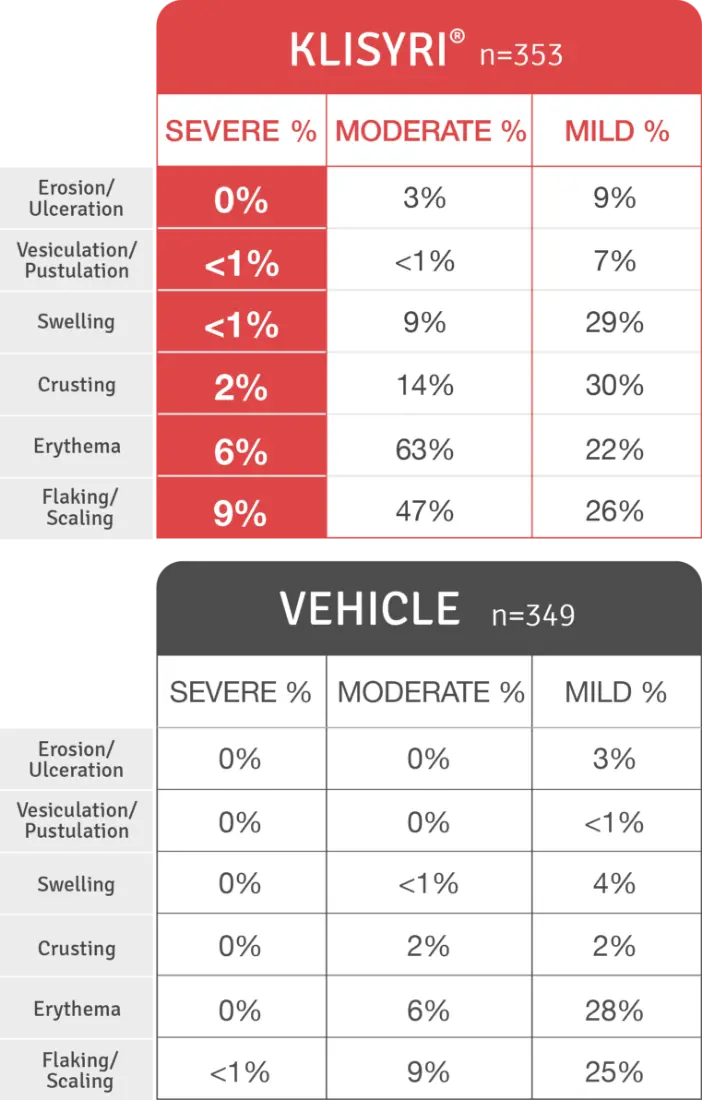
No patients withdrew from the trials due to adverse reactions1
TAP TO ENLARGE
SCALP
SCALP
CLICK OR TAP TO ENLARGE
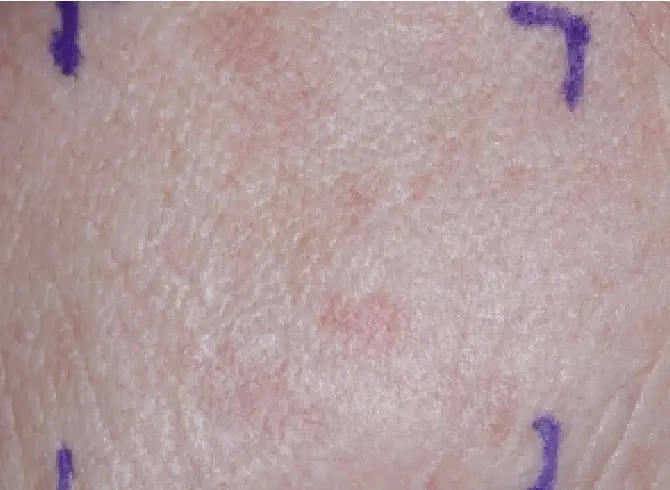
TAP TO ENLARGE
FACE
FACE
CLICK OR TAP TO ENLARGE
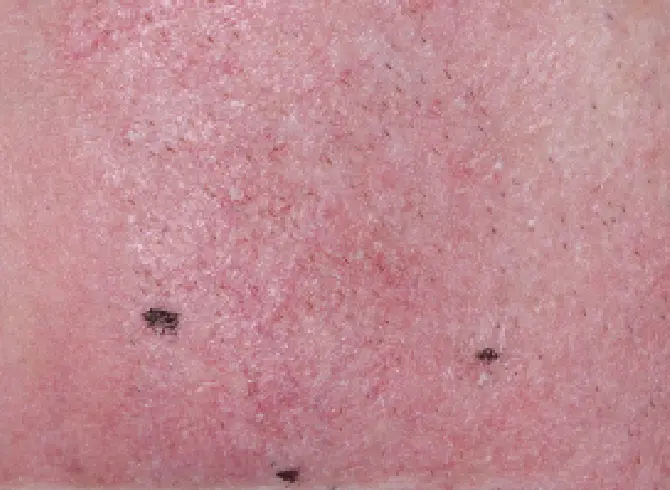
TAP TO ENLARGE
FACE
FACE
CLICK OR TAP TO ENLARGE
Clinical studies in healthy subjects demonstrated KLISYRI® did not cause contact sensitization (n=261), phototoxic skin reactions (n=31), or photoallergic skin reactions (n=64)1
The mean LSR composite score after treatment with KLISYRI® peaked on day 8, rapidly decreased by day 15, and resolved by day 292,3
†
LSRs were assessed using a grading scale: 0=absent, 1=mild (slightly, barely
perceptible), 2=moderate (distinct presence), and 3=severe (marked, intense). The LSR
composite score was the sum of the grades of LSRs at each visit (0 to
18).1
LSRs: local skin reactions.
KLISYRI is a microtubule inhibitor indicated for the topical treatment of actinic keratosis of the face or scalp.
KLISYRI may cause eye irritation. Avoid transfer of the drug into the eyes and to the periocular area during and after application. Wash hands immediately after application. If accidental exposure occurs, instruct patient to flush eyes with water and seek medical care as soon as possible.
Local skin reactions, including severe reactions (erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration) in the treated area can occur after topical application of KLISYRI. Avoid use until skin is healed from any previous drug, procedure, or surgical treatment. Occlusion after topical application of KLISYRI is more likely to result in irritation.
The most common adverse reactions (incidence ≥2%) were local skin reactions, application site pruritus, and application site pain.
To report an adverse event or product complaint, call or email: Medical Affairs and Customer Relations • Phone: 1-866-665-2782 • Fax: 510-595-8183 • Email: almirallmc@eversana.com
KLISYRI is a microtubule inhibitor indicated for the topical treatment of actinic keratosis of the face or scalp.
KLISYRI may cause eye irritation. Avoid transfer of the drug into the eyes and to the periocular area during and after application. Wash hands immediately after application. If accidental exposure occurs, instruct patient to flush eyes with water and seek medical care as soon as possible.
Local skin reactions, including severe reactions (erythema, flaking/scaling, crusting, swelling, vesiculation/pustulation, and erosion/ulceration) in the treated area can occur after topical application of KLISYRI. Avoid use until skin is healed from any previous drug, procedure, or surgical treatment. Occlusion after topical application of KLISYRI is more likely to result in irritation.
The most common adverse reactions (incidence ≥2%) were local skin reactions, application site pruritus, and application site pain.
To report an adverse event or product complaint, call or email: Medical Affairs and Customer Relations • Phone: 1-866-665-2782 • Fax: 510-595-8183 • Email: almirallmc@eversana.com
References:
1. KLISYRI® [package insert]. Malvern, PA: Almirall, LLC, 2021. 2. Data on file. 2019. KX01-AK-004. 3. Data on file. 2019. KX01-AK-003.